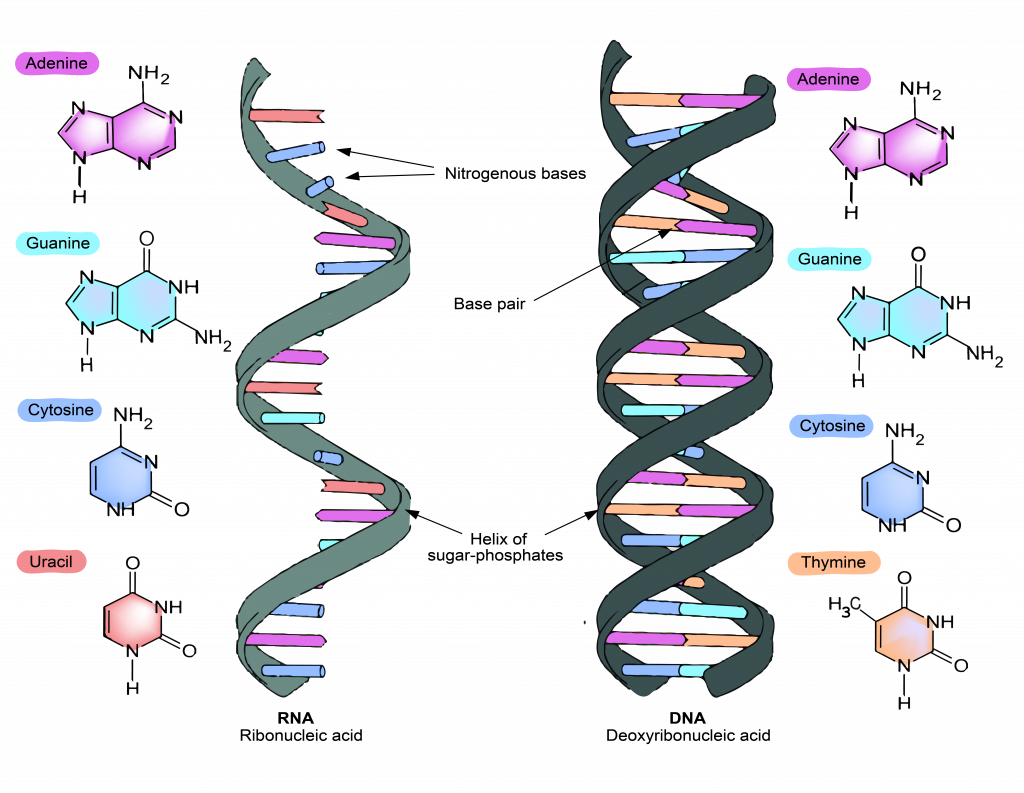
Stacking interactionsĭNA has an interesting arrangement wherein the non-polar, uncharged bases are present in the interior of the structure, while the negatively charged phosphates are present on the outside. The width of each of these ‘rungs’ are the same as it involves one purine (A or G) and one pyrimidine (C or T) base. The sugar-phosphate chains form the backbone of the ladder-like DNA structure and these base pairs form the rungs. A-T pair forms two hydrogen bonds, while C-G pair forms three. This selective pairing is called ‘complementary base pairing’. This pairing is very specific: adenine pairs with thymine and cytosine pairs with guanine. Although individually each hydrogen bond is much weaker than the covalent bond, they can stabilize the double helix because of their large numbers. Hydrogen bonds occur over short distances and can be easily formed and broken. There is no exchange or sharing of electrons in hydrogen bonds as seen in covalent or ionic bonds. The hydrogen bonds between the base pairs form the double helical structure of DNA. This is formed between the 5’-phosphate group of one nucleotide and the 3’-OH group of another nucleotide forming a sugar-phosphate backbone of DNA. This addition involves formation of a covalent bond called the ‘phosphodiester bond ’. New high-throughput genomic array offers an in-depth view of the mouse epigenome.Study calls for more conservation efforts to preserve adaptive DNA diversity of dolphin populations.A different approach to build a more durable, broadly effective COVID-19 vaccine.
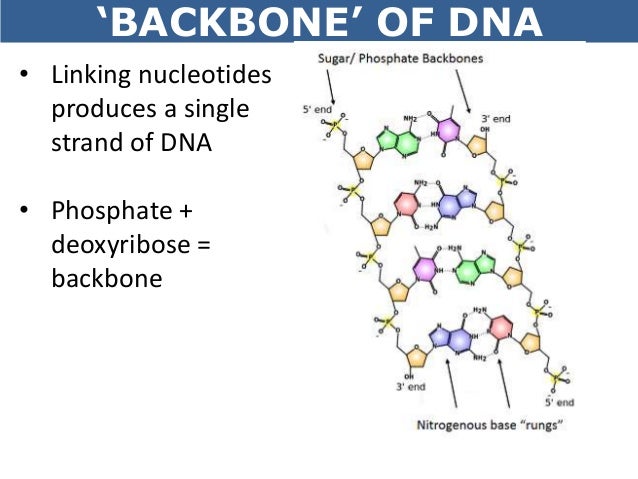
However, when a nucleotide is incorporated into a DNA strand, it loses two of the phosphate groups and only one phosphate group is added to the DNA strand.
Dna backbone structure free#
In an unattached and free nucleotide, there is a triphosphate group on the 5’-carbon of the deoxyribose sugar. Each of these bases is connected to 1’-carbon of the deoxyribose sugar. Thus, there are four different nucleotides that can be incorporated into DNA.īased on which base is attached, the nucleotides are called 2’-deoxyadenosine triphosphate, 2’-deoxycytidine triphosphate, 2’-deoxyguanosine triphosphate, or 2’-deoxythymidine triphosphate. Purines have two carbon-nitrogen rings while pyrimidines have a single carbon-nitrogen ring. There are four bases: Adenine and Guanine (purines) Cytosine and Thymine (Pyrimidines).
Reviewed by Deepthi Sathyajith, M.Pharm.ĭNA consists of two strands, that wind around each other.
